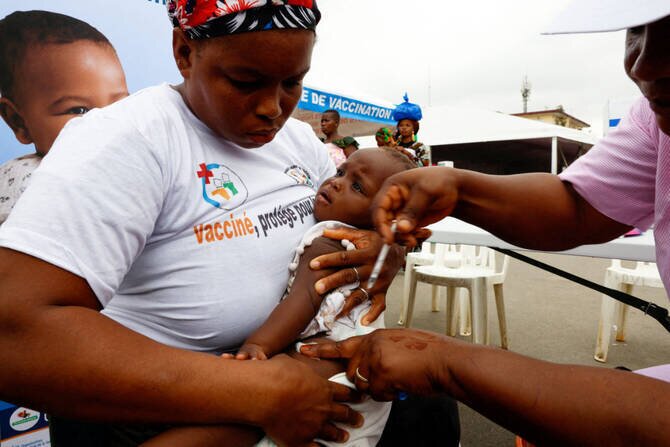
WHO Approves First Malaria Treatment for Infants
Artemether-lumefantrine gains prequalification approval to combat malaria in newborns and infants.
The World Health Organization (WHO) announced on Friday the prequalification approval of artemether-lumefantrine, a novel antimalarial formulation designed specifically for newborns and infants.
This milestone marks the first time the WHO has given such approval for a malaria treatment targeting the youngest victims of the mosquito-borne disease.The designation indicates that the medicine meets international standards of quality, safety, and efficacy according to the WHO.
Previously, infants were treated with formulations intended for older children, which presented a higher risk of dosage errors, side effects, and toxicity.
This new development aims to address these issues by providing tailored treatment for infants.WHO Director-General Tedros Adhanom Ghebreyesus emphasized the significant impact malaria has had on communities, but highlighted the changing narrative with new vaccines, diagnostic tests, next-generation mosquito nets, and effective medicines adapted for young children.
He stressed that ending malaria is now a real possibility but requires sustained political and financial commitment.In 2024, there were an estimated 282 million malaria cases and 610,000 deaths globally in 80 countries, predominantly affecting Africa with 95% of cases and deaths.
Children under five account for three-quarters of these deaths.
The WHO attributes the progress against malaria to drug resistance, insecticide resistance, diagnostic failures, and decreased foreign aid spending.The prequalification process enables public sector procurement, aiming to close a treatment gap for approximately 30 million babies born annually in malaria-endemic areas of Africa.
The WHO notes that 70% of countries lack robust regulatory systems necessary for overseeing medicines, vaccines, tests, and medical devices.
Therefore, the prequalification program ensures key health products meet global standards of quality, safety, efficacy, and performance before they are procured internationally.
This milestone marks the first time the WHO has given such approval for a malaria treatment targeting the youngest victims of the mosquito-borne disease.The designation indicates that the medicine meets international standards of quality, safety, and efficacy according to the WHO.
Previously, infants were treated with formulations intended for older children, which presented a higher risk of dosage errors, side effects, and toxicity.
This new development aims to address these issues by providing tailored treatment for infants.WHO Director-General Tedros Adhanom Ghebreyesus emphasized the significant impact malaria has had on communities, but highlighted the changing narrative with new vaccines, diagnostic tests, next-generation mosquito nets, and effective medicines adapted for young children.
He stressed that ending malaria is now a real possibility but requires sustained political and financial commitment.In 2024, there were an estimated 282 million malaria cases and 610,000 deaths globally in 80 countries, predominantly affecting Africa with 95% of cases and deaths.
Children under five account for three-quarters of these deaths.
The WHO attributes the progress against malaria to drug resistance, insecticide resistance, diagnostic failures, and decreased foreign aid spending.The prequalification process enables public sector procurement, aiming to close a treatment gap for approximately 30 million babies born annually in malaria-endemic areas of Africa.
The WHO notes that 70% of countries lack robust regulatory systems necessary for overseeing medicines, vaccines, tests, and medical devices.
Therefore, the prequalification program ensures key health products meet global standards of quality, safety, efficacy, and performance before they are procured internationally.










